$20.00
Description
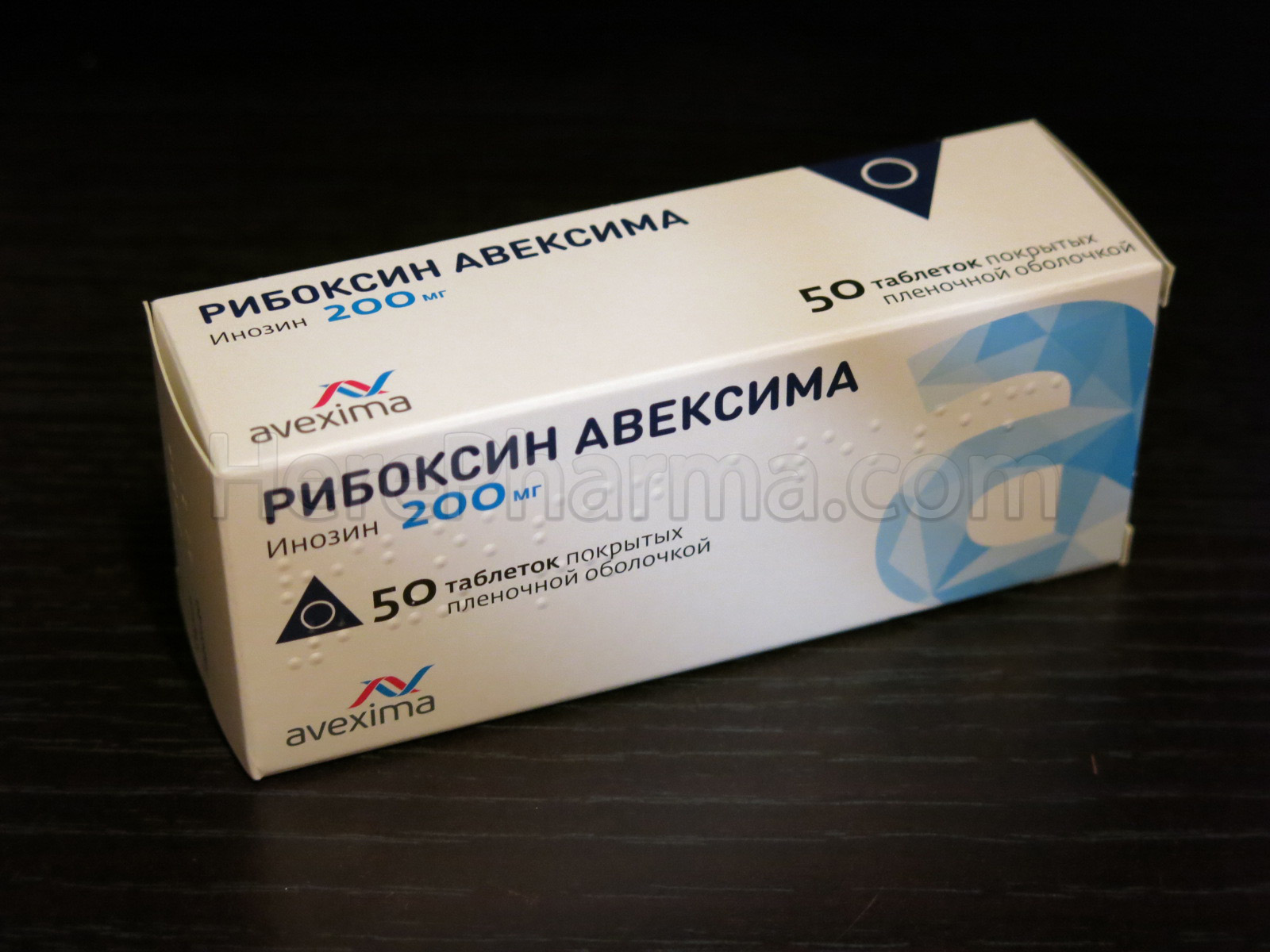
Riboxine (Inosine) 200mg 50 tablets
Pharmacological action
Nucleoside purine, a precursor of ATP. It improves myocardial metabolism, has anti-hypoxic and antiarrhythmic effects. Increases the energy balance of the myocardium. It has a protective effect on the kidneys in ischemia during surgery.
Participates in glucose metabolism, increases the activity of a number of enzymes of the Krebs cycle. Stimulates the synthesis of nucleotides.
Pharmacokinetics
Inosine is well absorbed from the gastrointestinal tract. It is metabolized in the liver to form glucuronic acid and its subsequent oxidation. In small amounts excreted by the kidneys.
Indications
IHD, myocardiodystrophy, condition after myocardial infarction, congenital and acquired heart defects, cardiac arrhythmias, especially with glycoside intoxication, myocarditis, dystrophic myocardial changes after heavy physical exertion and infectious diseases or due to endocrine disorders; hepatitis, liver cirrhosis, fatty liver dystrophy, including those caused by alcohol or drugs; prevention of leukopenia with radioactive irradiation; surgery on an isolated kidney (as a means of pharmacological protection in the temporary absence of blood circulation in the operated organ).
Dosage regimen
When taken orally, the initial daily dose is 600-800 mg, then the dose is gradually increased to 2.4 g/day in 3-4 doses.
When/in (jet or drip) the initial dose is 200 mg 1 time / day, then the dose is increased to 400 mg 1-2 times / day.
The duration of the course of treatment is set individually.
Side effect
Possible: itching, redness of the skin.
Rare: increased uric acid concentration.
Contraindications to use
Gout, hyperuricemia, hypersensitivity to inosine.
Use in renal impairment
With caution, inosine is prescribed for violations of kidney function.
Special instruction
With caution, inosine is prescribed for violations of kidney function. When itching and hyperemia of the skin inosine should be abolished.